- Marcos Santos, Ph.D
- 0 Comments
- 198 Views
Molecular tests should be performed after fine-needle aspiration biopsy (FNA).
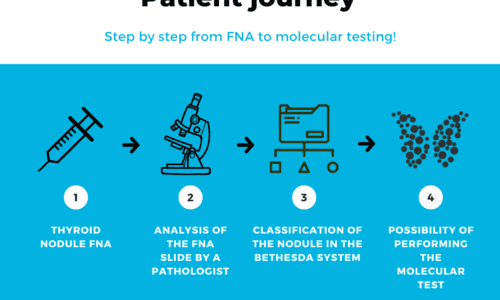
The patient’s journey with a thyroid nodule often involves undergoing FNA. The responsible physician (usually an endocrinologist or head and neck surgeon) may recommend the biopsy based on certain characteristics of the nodule observed on ultrasound that raise suspicion. FNA is an important exam for a more detailed analysis of the nodule’s condition.
After the biopsy, it is classified according to the standardized Bethesda system(1). The classification varies based on the characteristics of the nodule cells and can be classified from I to VI as follows:
I – Non-diagnostic sample (insufficient)
II – Benign
III – Atypia of undetermined significance / follicular lesion of undetermined significance
IV – Suspicious for follicular neoplasm – undetermined
V – Suspicious for malignancy
VI – Malignant
This is where molecular tests can help. The Bethesda III and IV classifications are considered indeterminate, meaning that the characteristics of the biopsy do not clearly indicate whether the nodule is benign or malignant, leaving room for doubts and uncertainties about the nodule. The standard procedure, especially when dealing with a possible tumor, is to remove the nodule through surgery. However, in about 80% of cases, the analysis of the nodule after thyroid removal shows that the nodule was benign(2), and that surgery was unnecessary. This is where the mir-THYpe full molecular test can help.
How does the mir-THYpe full molecular test work?
The nodule is a mass of abnormally growing cells. The molecular test evaluates whether there are any molecular (“genetic”) characteristics in these nodule cells that indicate whether it is possibly malignant or benign. This is made possible through Molecular Biology techniques and sophisticated Artificial Intelligence analysis.
With this algorithm, the molecular test can reclassify the nodule that was originally indeterminate as either positive or negative for malignancy with high sensitivity and specificity, providing a more accurate diagnosis, avoiding surgeries, and helping with the feeling of uncertainty that many patients have when they receive the result of an indeterminate nodule (Bethesda III or IV).
What should be done when the nodule is reclassified as potentially malignant?
Some specific molecular markers that suggest the nodules have the potential to be more or less aggressive are already known in the literature. These are called prognostic markers(3, 4). The mir-THYpe full also analyzes these markers. So, if the test indicates that the nodule is potentially malignant, it will also show whether it has any of these markers of aggressiveness or surgical urgency, providing the physician with a more precise and personalized perspective on each patient and the surgery, including how it needs to be performed and with what urgency.
If the FNA is classified as Bethesda V or VI, can molecular tests help me in this case?
Yes, molecular tests that assess tumor markers of aggressiveness, urgency, and surgical extent can be used for the physician to have a better interpretation of the patient’s case and provide a more personalized surgery. Onkos has developed the mir-THYpe pre-op specifically for these cases. Its purpose is to evaluate the genetic markers of nodules with a surgical indication (Bethesda V or VI) to understand if the nodule is more aggressive and requires a more extensive surgery. In other words, it offers support for a more personalized decision-making process for the physician during this delicate moment for the patient.
But do both tests do the same thing?
No, mir-THYpe full is indicated for Bethesda III or IV nodules because it allows the reclassification of indeterminate nodules as potentially benign or malignant. It also analyzes prognostic markers, which are markers of aggressiveness and surgical urgency.
On the other hand, mir-THYpe pre-op is indicated for Bethesda V or VI nodules that have a surgical indication. It only includes the prognostic markers since the surgery will already be performed, and the patient does not require the diagnostic reclassification of the nodule. Therefore, it provides support to the physician in performing a more personalized surgery for each patient!
References:
- The 2017 Bethesda System for Reporting Thyroid Cytopathology | Thyroid- https://www.liebertpub.com/doi/full/10.1089/thy.2017.0500
- Bongiovanni M, et al. 2012 Acta Cytologica
- Haugen BR, et al., 2016 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26: 1–1333
- Bittar GC, et al., 2019 Painel sérico de microRNAs (MiRs) como método diagnóstico e prognóstico em pacientes com nódulos de Tireoide. Arch Endocrinol Metab 63 (Sup) Abst PT.170